
Ordinary copper sulfate, for example, normally occurs in a configuration that includes four water molecules surrounding a single copper ion. For example, they tend to form complexes, compounds in which a group of atoms cluster around a single metal atom. In addition, the transition metals share some chemical properties. They also have a shiny, lustrous, metallic appearance that may range from silver to gold to white to gray. With the notable exception of mercury, the only liquid metal, they all have relatively high melting and boiling points. The transition elements share many physical properties in common. The discoverers of technetium were Italian physicists Carlo Perrier and Emilio Segr é (1905 –1989). It was originally produced synthetically in 1937 among the products of a cyclotron reaction. Technetium (element #43) is one of only two "light" elements that does not occur in nature. One member of the transition family deserves special mention. 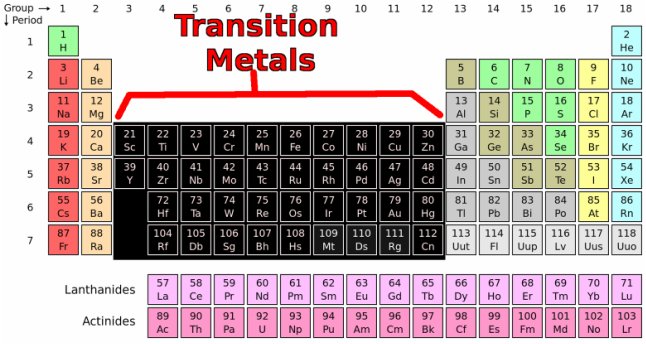
These elements include titanium, vanadium, manganese, zirconium, molybdenum, palladium, and tungsten. A number of other transition elements are probably somewhat less familiar, although they have vital industrial applications. These elements, all of which are metals, include some of the best-known names on the periodic table -iron, gold, silver, copper, mercury, zinc, nickel, chromium, and platinum among them. The transition elements are the elements that make up Groups 3 through 12 of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
